Acidity is one of the most important — and most misunderstood — elements in beverage development.
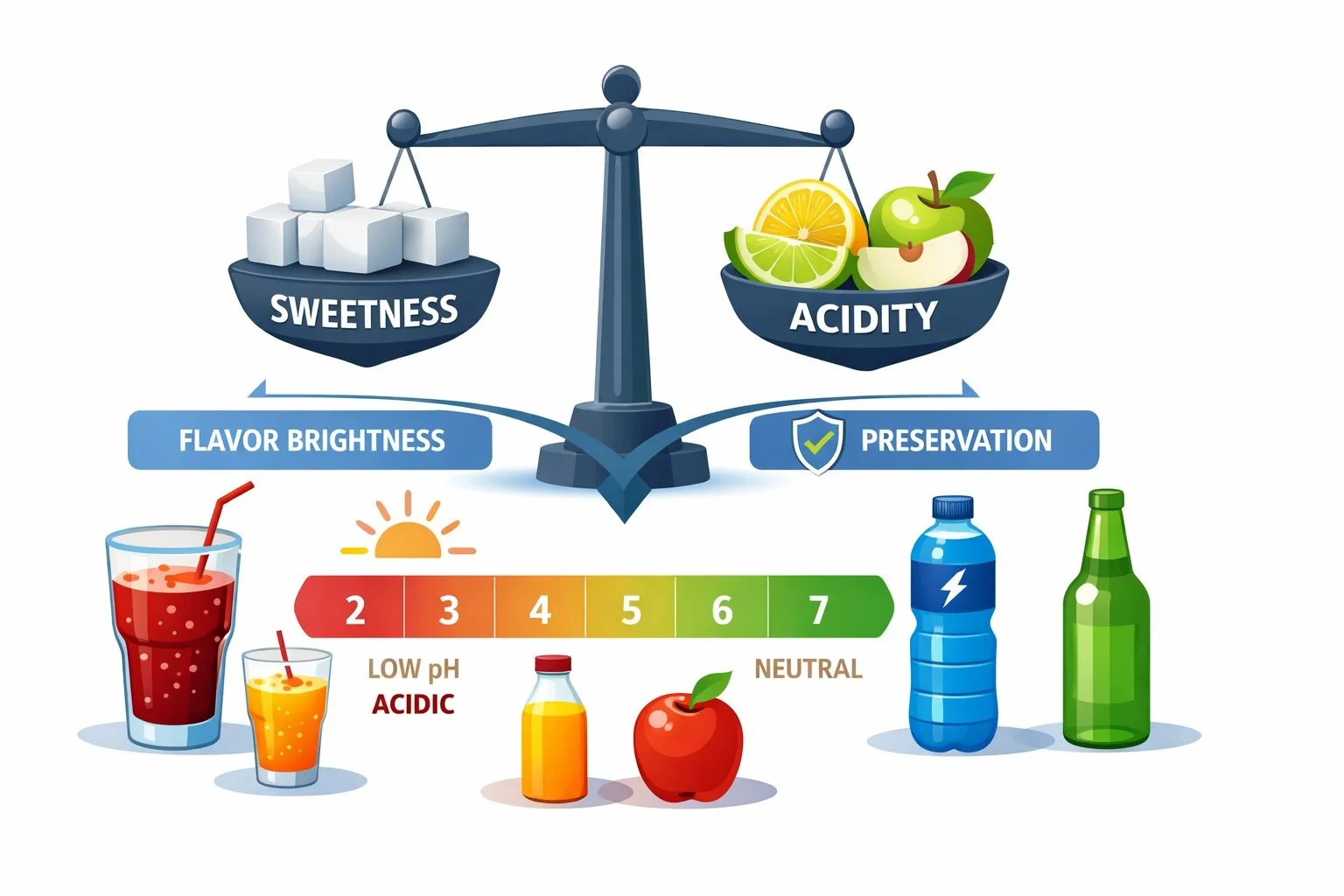
When people think about acidity, they often associate it simply with sourness. In reality, acidity does much more than make a drink taste tart. It shapes flavor brightness, balances sweetness, enhances refreshment, and plays a major role in shelf stability.
In fact, many beverages would taste flat or overly sweet without the presence of acids.
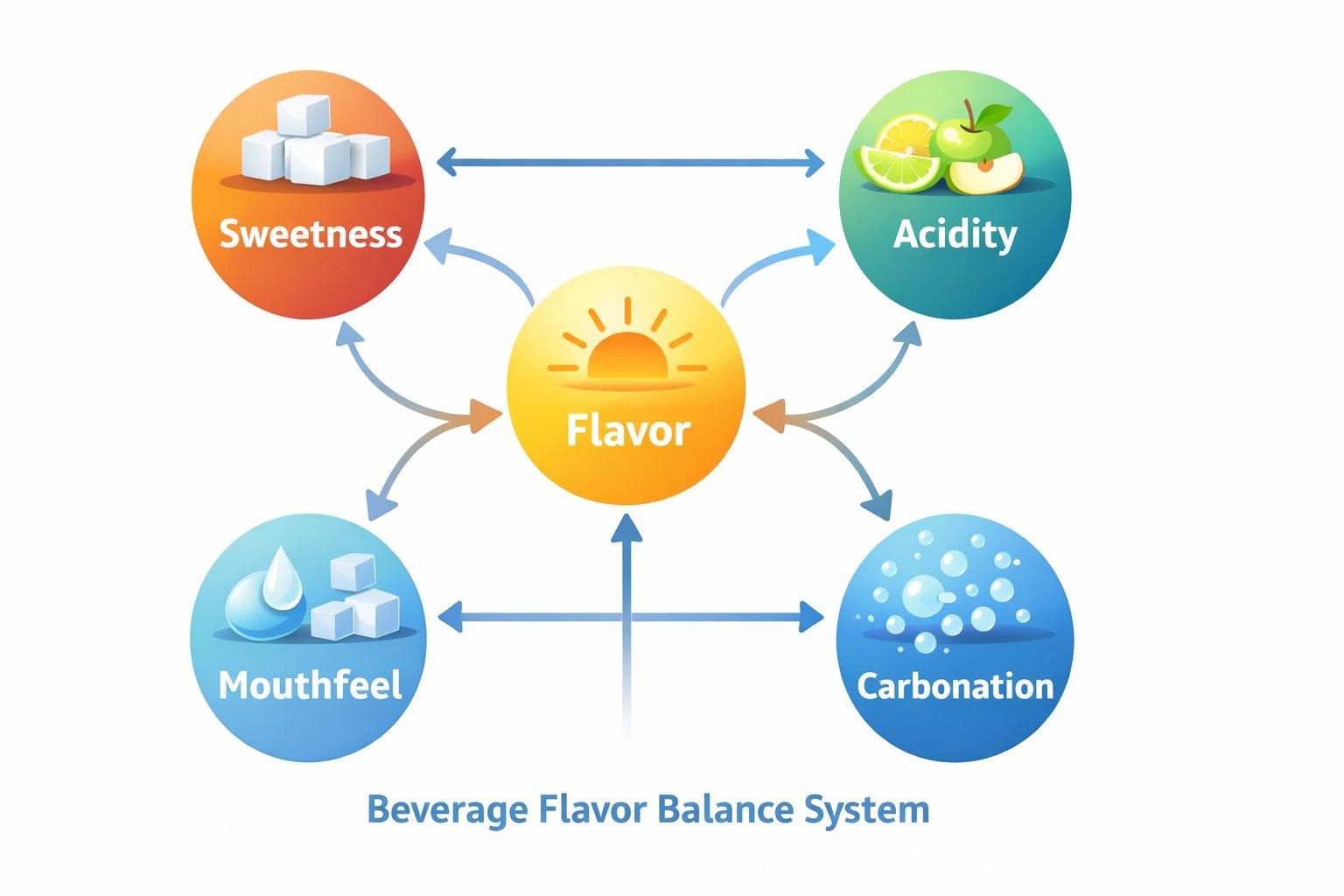
Acids also influence preservation systems, microbial stability, ingredient functionality, and even manufacturing decisions. Because of these wide-ranging effects, acidity is one of the core formulation systems beverage developers adjust when building a drink.
Understanding how acid systems work helps founders create beverages that taste vibrant, balanced, and stable over time.
This is why acidity sits alongside sweeteners, flavors, carbonation, and preservation as one of the core building blocks of beverage formulation. As discussed in the Power Brands introduction guide, these formulation systems interact closely and must be balanced together rather than treated separately.
Why This Matters in Beverage Development
-
Flavor Balance
Acidity counteracts sweetness and prevents beverages from tasting heavy or syrupy. Many fruit flavors feel brighter and more refreshing when paired with the right acid levels.
Flavor Expression
Acids can enhance flavor perception. Citrus, berry, and tropical flavors often become more vibrant when acidity is properly balanced.
Shelf Stability
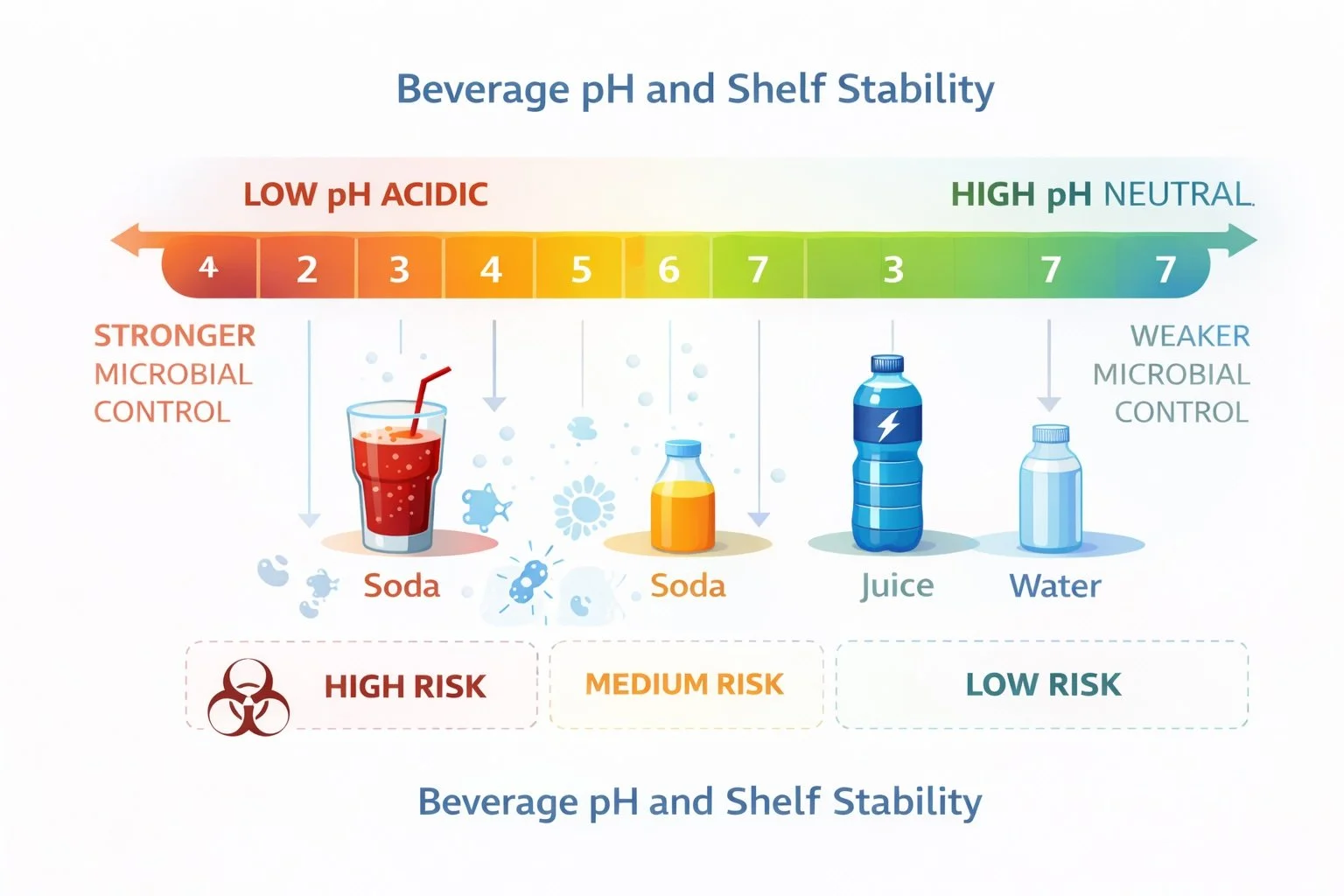
Acidity directly affects microbial stability. Lower pH environments inhibit microbial growth, which is why many shelf-stable beverages rely on acid systems as part of their preservation strategy.
Preservatives like sodium benzoate and potassium sorbate are also more effective in lower pH environments, meaning acid systems often work together with preservation strategies.
Mouthfeel and Drinkability
Acidity contributes to the refreshing sensation many consumers expect from beverages. It creates a crisp, clean finish that encourages repeat drinking.
Carbonated Beverage Experience
Carbonation increases the perception of acidity, which is why formulators often adjust acid levels when designing sparkling drinks.
Core Principle
The most important insight about beverage acidity is this:
Acids are used primarily to balance sweetness and enhance flavor — not just to create sourness.
Beginner assumption:
Acids are added to make a drink sour.
Industry reality:
Acids are used to create balance, brightness, and stability.
In most beverages, acidity acts like a structural component that shapes the overall flavor experience.
Think of acidity as the element that keeps a beverage lively. Without it, drinks tend to taste dull, overly sweet, or heavy.
Main Approaches
Beverage developers typically work with a small group of acids that each provide slightly different sensory characteristics.
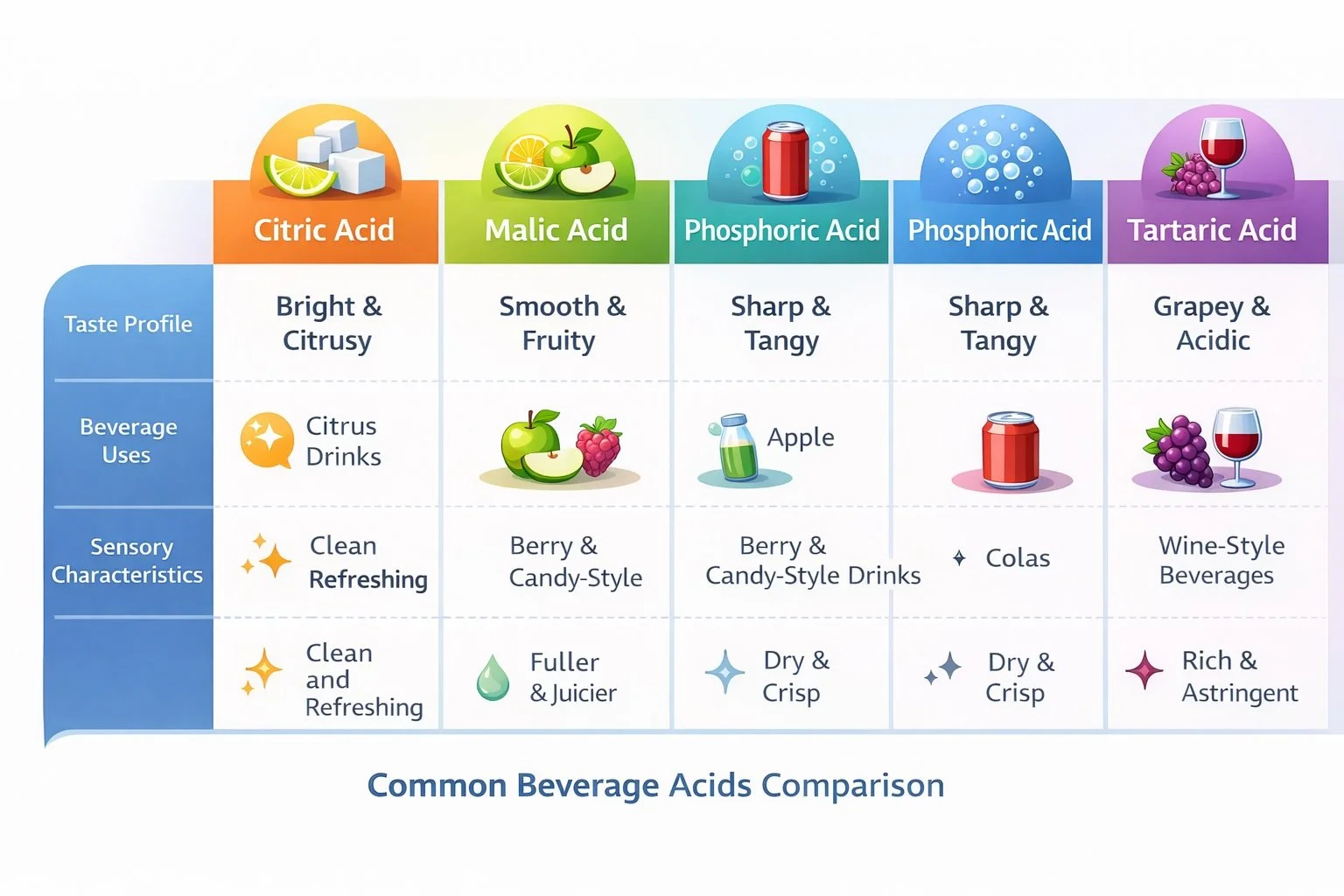
Citric Acid
Citric acid is the most widely used acid in beverages.
It is commonly associated with citrus fruits and provides a clean, bright sourness that pairs well with fruit flavors.
Citric acid is used heavily in:
• soft drinks
• sports drinks
• energy drinks
• flavored waters
• fruit beverages
Because of its versatility and cost effectiveness, it is often the default acid used in beverage formulation.
Malic Acid
Malic acid produces a smoother, longer-lasting sourness.
It is naturally associated with apples and is often used to create fuller fruit flavor experiences.
Malic acid works especially well in:
• apple flavors
• berry beverages
• candy-style drinks
• energy drinks
Many beverage developers combine citric and malic acid to create layered acidity.
Phosphoric Acid
Phosphoric acid is widely used in cola beverages.
It produces a sharper, slightly drier acidity compared to fruit acids.
This acid helps create the distinctive taste profile associated with many dark sodas.
Tartaric Acid
Tartaric acid is commonly associated with grapes and wine.
It is less frequently used in mainstream beverages but can be important in grape drinks, wine-based beverages, and certain specialty products.
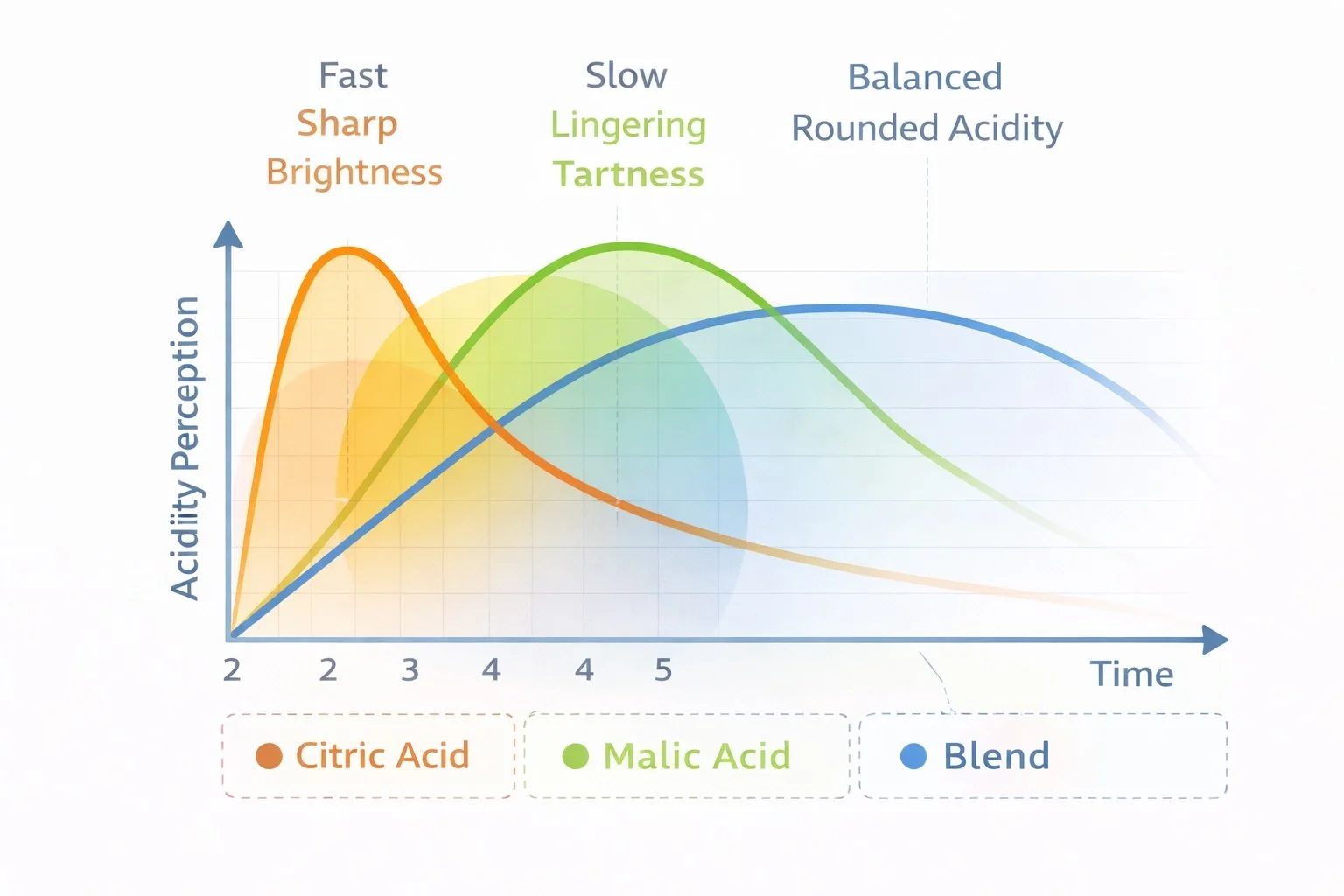
Blended Acid Systems
Many beverages use acid blends rather than a single acid.
Blending allows developers to fine-tune the shape of acidity:
• immediate brightness
• lingering tartness
• smoother finish
Just like sweeteners are often blended to create better taste profiles, acids are frequently combined to produce a more natural flavor experience.
This system-based thinking is common throughout beverage formulation. For example, sweetness systems are also often blended to shape flavor perception and balance.
How This Affects the Formula
Acidity affects several parts of the beverage formula simultaneously.
Taste Balance
Acid levels must be adjusted relative to sweetness.
Higher sweetness usually requires higher acidity to maintain balance.
Flavor Perception
Acids amplify certain flavor notes.
For example:
• citrus flavors become brighter
• berry flavors feel juicier
• tropical flavors feel fresher
This is why acidity is considered part of the flavor system, not just a standalone ingredient.
pH and Preservation
Acid levels determine the pH of the beverage.
Most shelf-stable beverages fall between:
pH 2.5 – 4.0
This range helps control microbial growth and supports preservation systems.
Mouthfeel
Acidity influences how refreshing a beverage feels.
High acidity → crisp and refreshing
Lower acidity → smooth and mellow
Cost
Acids are relatively inexpensive ingredients, but small adjustments can significantly affect the flavor balance of the beverage.
This means acidity often becomes one of the final tuning tools used during formulation.
Common Beginner Mistakes
Treating Acidity as an Afterthought
Acid balance should be developed alongside sweetness and flavor — not added at the end.
Using Too Much Acid
New developers sometimes increase acid levels to create “more flavor.”
This often produces a harsh or unpleasant sourness.
Ignoring Acid Blends
Using only one acid can create a flat acidity profile.
Blending acids often produces a more natural taste.
Not Considering pH
Acid choices affect microbial stability and preservation strategies.
Ignoring pH can create shelf-life problems later.
Practical Advice for Founders
When developing your beverage, consider these principles:
1. Balance sweetness and acidity together
These two systems must always be adjusted in tandem.
2. Taste with different acid levels
Small adjustments can dramatically change drinkability.
3. Consider acid blends
Citric + malic is one of the most common combinations.
4. Measure pH early
pH affects both safety and preservation.
5. Test with carbonation if applicable
Carbonation increases perceived acidity, which may require adjustments.
Key Takeaway
Acidity is one of the foundational building blocks of beverage formulation.
It shapes flavor brightness, balances sweetness, improves drinkability, and supports preservation systems.
When used correctly, acids help transform a beverage from flat and overly sweet into something vibrant, refreshing, and stable.
For beverage founders, understanding acid systems is essential to creating drinks that taste great and perform well in the real world.